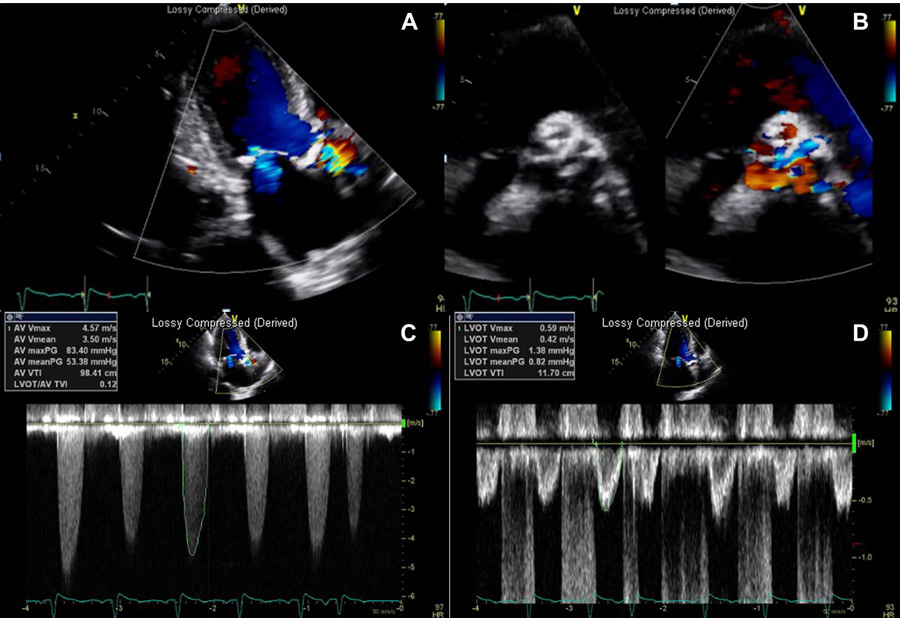
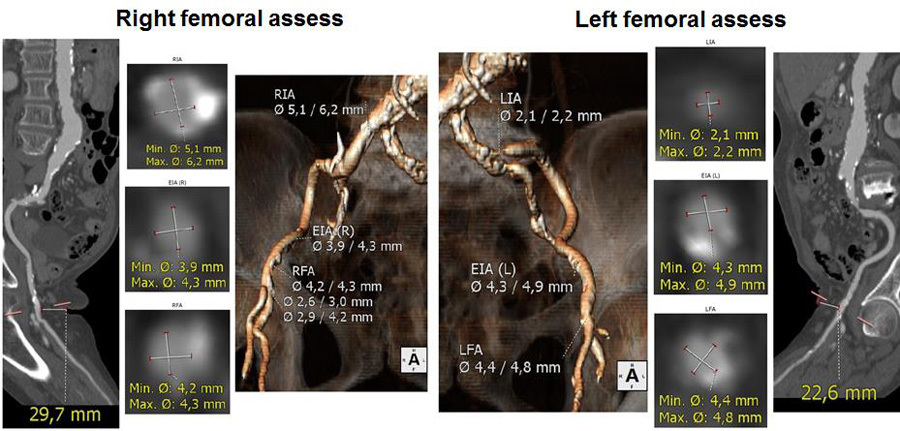
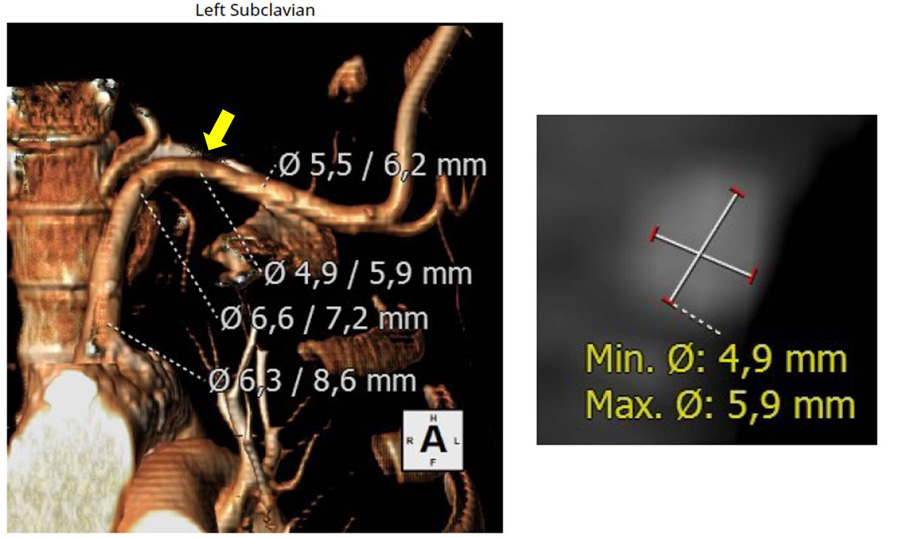
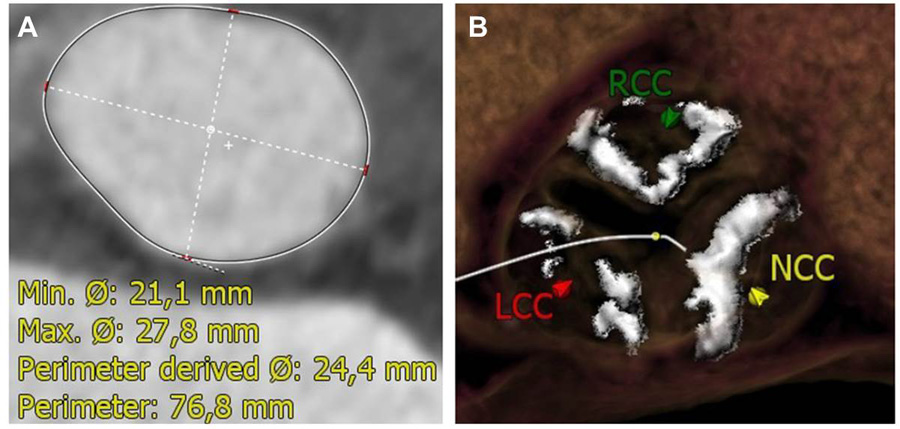
A 78 year-old woman visited Chonnam National University Hospital because of dyspnea and chest tightness which had developed 2 years ago. She had histories of hypertension, and myocardial infarction treated percutaneous coronary intervention for left anterior descending (LAD) artery lesion. On physical exam, systolic ejection murmur was heard on right upper sternal border. Twelve-lead electrocardiogram (ECG) showed normal sinus rhythm, left ventricular hypertrophy, and ST-segment depression on lateral leads which indicated left ventricular hypertrophy strain pattern. Transthoracic echocardiography revealed severe aortic valve stenosis (AS) with peak velocity through the aortic valve was 4.57 m/sec, the mean pressure gradient was 53.38 mmHg, and aortic valve area was 0.41 cm2 calculated by continuity equation (Figure 1). The aortic valve replacement was required, so surgical risk was assessed. The Society of Thoracic Surgeons score (STS) was 8.951% and the European System for Cardiac Operative Risk Evaluation (EuroSCORE) II was 18.54% which indicated high risk for operation. The patient and her protectors did not agree to take a surgical management, so transcatheter aortic valve implantation (TAVI) was planned. Coronary angiography and aortography was performed as pre-evaluations for TAVI. Angiography showed diffuse atherosclerotic lesions, but no in-stent restenosis in proximal LAD stent, or no significant stenosis over both coronary arteries, but tortuous and stenosis with severe calcification over both iliofemoral arteries. The diameters of both iliofemoral arteries were less than 5 mm on computed tomography (CT) scan (Figure 2). To find alternative assess, upper extremities CT scan was checked and it showed a focal stenosis in proximal left subclavian artery, but general diameters were around 6 mm (Figure 3). It could be possible to do TAVI procedure via axillary approach. The perimeter at aortic annulus was 76.8 mm and there was severe calcification at 3 aortic valve cusps (Figure 4). Therefore, 29 mm-diameter self-expandable Evolut R® was selected.
Under general anesthesia, TAVI was started under guidance of transesophageal echocardiography (TEE) and fluoroscopy. After surgical cut-down at left axillary artery, the sheath placed in ascending aorta (Figure 5). A 29 mm-diameter Evolut R® was inserted with a temporary pacemaker kept on (Figure 6) Follow-up aortography and TEE revealed mild paravalvular leakage (Figure 7). Initial peak-to-peak pressure gradient between aorta and left ventricle was 60 mmHg and post-procedural pressure was markedly decreased. After the aortic valve implantation, there was leaner tear on subclavian artery where sheath was inserted. Angioplasty was performed using bovine pericardium.
After successful TAVI, follow-up echocardiography showed no significant paravalvular leakage and good apposition of artificial valve. Peak aortic valve velocity was decreased to 2.53 m/sec (Figure 8). The patient was discharged without other complications.
Aortic valve replacement still has been the mainstay treatment of severe AS. In large clinical studies, the results of TAVI procedure were comparable to those of surgical valve replacement. The number of TAVI procedures is increasing rapidly, and the role of TAVI is evolving. The replacement valve is delivered via four types of access methods: transfemoral, subclavian (axillary), direct aortic, and transapical. Usually, femoral access is preferred for no need of surgical cut-down. If patient is not suitable for femoral approach due to atherosclerosis, calcifications, or tortuosity, altanative route could be selected. Subclavian arteries are often viable in patients with compromised iliofemoral arteries. Subclavian access also often presents a better access angle for patients with extremely angulated or horizontal anatomies. Some European countries have clinical experiences of subclavian approach and the outcomes of subclavian assess were comparable with those of transfemoral approach (74.6% vs. 72.1% in 2-year survival, Italian experience presented at TCT 2011). Therefore, subclavian access can be the 2nd best option for TAVI procedure for small incision size and possible local anesthesia.
 |
|
Figure 1. Transthoracic echocardiography revealed severe aortic valve stenosis (panel A and B) with peak velocity through the aortic valve was 4.57 m/sec (panel C), the mean pressure gradient was 53.38 mmHg, and aortic valve area was 0.41 cm2 calculated by continuity equation (panel D)
|
 |
|
Figure 2. The diameters of both iliofemoral arteries were less than 5 mm on computed tomography scan
|
 |
|
Figure 3. Upper extremities computed tomography scan showed a focal stenosis in proximal left subclavian artery (arrow), but general diameters were around 6 mm
|
 |
|
Figure 4. The perimeter at aortic annulus was 76.8 mm (panel A) and there was severe calcification at 3 aortic valve cusps (panel B) by computed tomography
|
 |
|
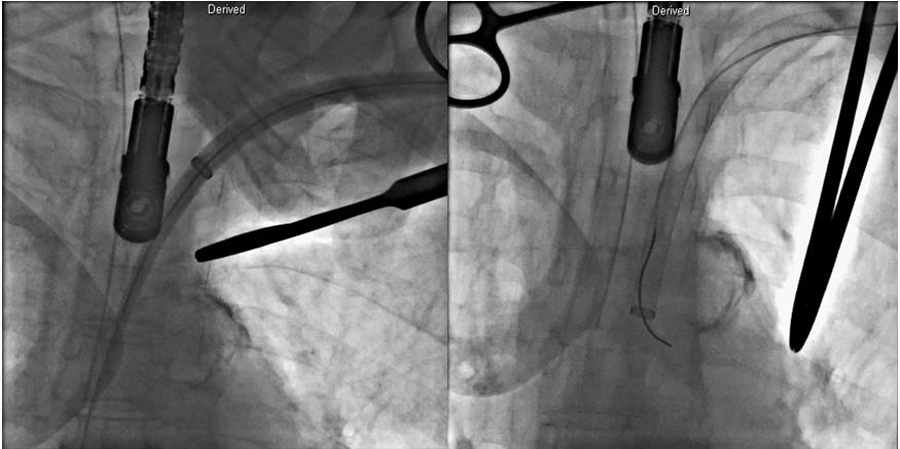
Figure 5. After surgical cut-down at left axillary artery, the sheath placed in ascending aorta
|
 |
|
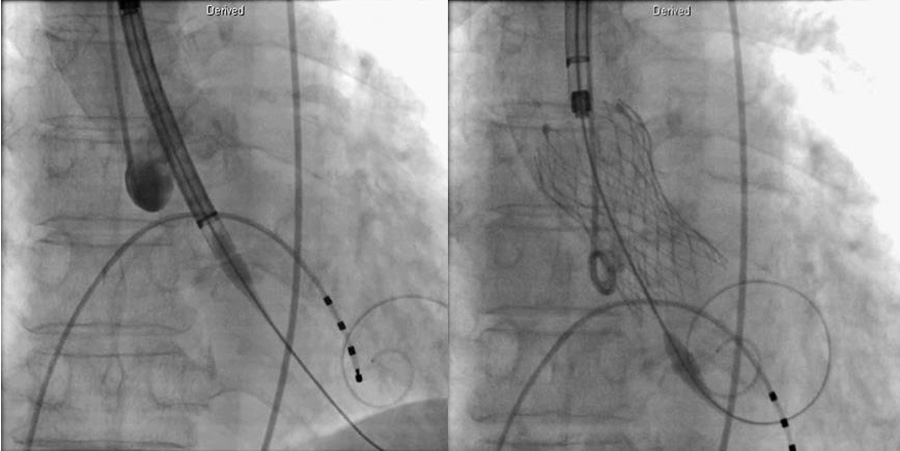
Figure 6. A 29 mm-diameter Evolut R® was inserted with a temporary pacemaker kept on via left subclavian approach
|
 |
|
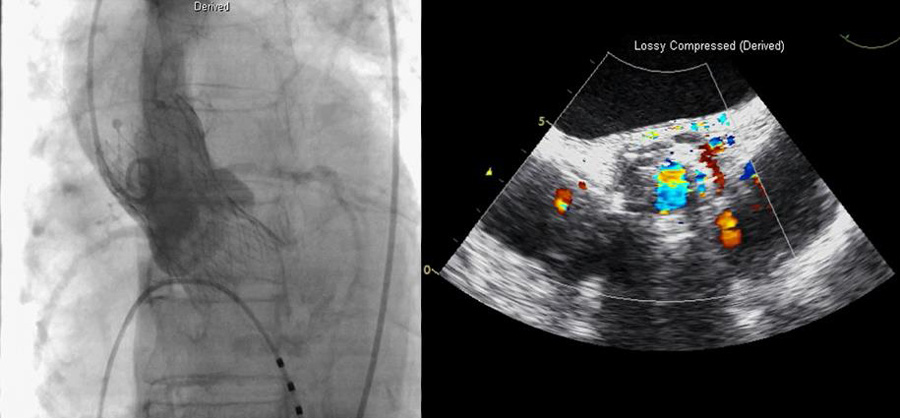
Figure 7. Follow-up aortography and transesophageal echocardiography revealed mild paravalvular leakage
|
 |
|
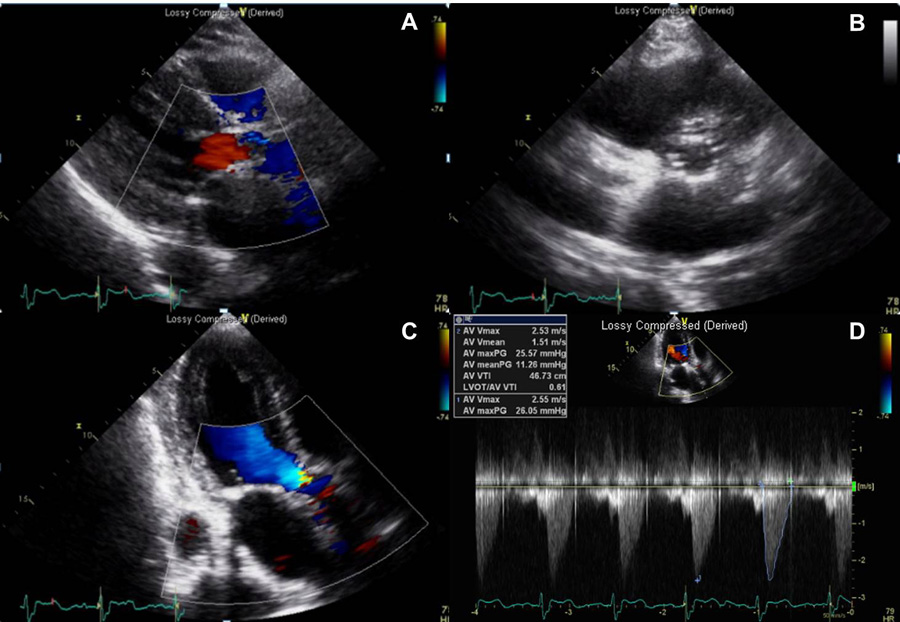
Figure 8. Follow-up echocardiography showed no significant paravalvular leakage (panel A and C) and good apposition of artificial valve (peak B). Peak aortic valve velocity was decreased to 2.53 m/sec (panel D)
|
|


















