Background
Percutaneous cardiopulmonary support (PCPS) using extracorporeal membrane oxygenation (ECMO) has been widely used to maintain cardiac and/or pulmonary functions in the patients with cardiopulmonary collapse. Almost all patients receive multiple and massive blood transfusion during the period of the ECMO application because of frequent laboratory monitoring and hemolysis. Especially when the ECMO circuit is removed, considerable blood volume loss cannot be avoidable owing to discarded blood in the ECMO circuit. If a patient does not accept the blood transfusion even at the risk of death due to any cause including his religious ground, the medical team cannot help taking up the challenge of clinical situation for the patient.
Case
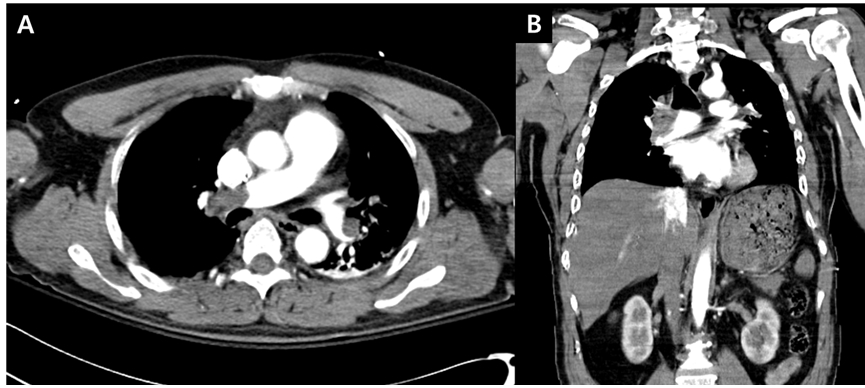
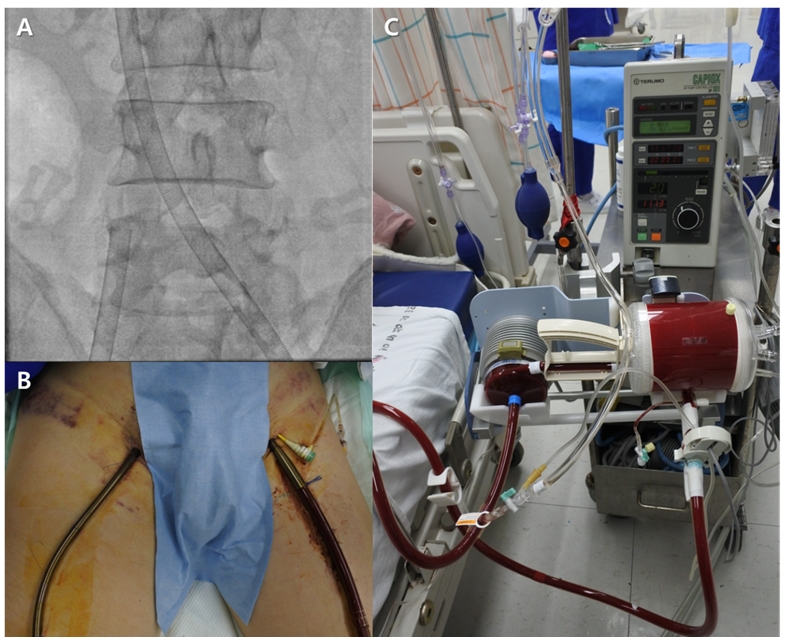
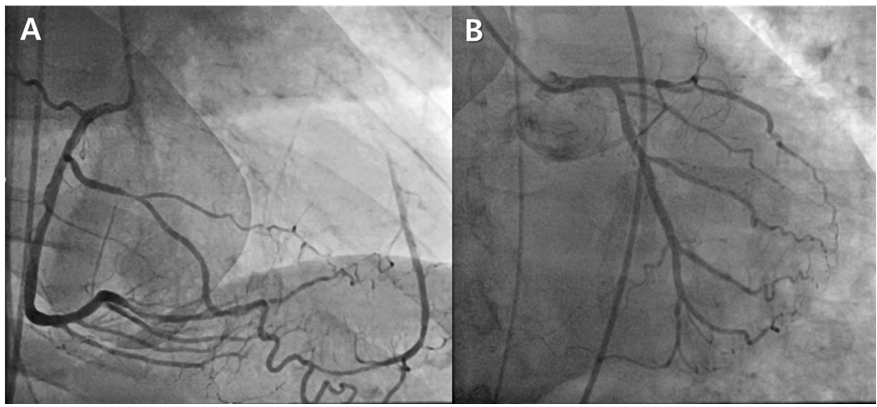
A 61-year-old male came to the emergency room (ER) complaining loss of consciousness and severe dyspnea. His family denied any special medical history such as hypertension and diabetes. In social history, his and his family’s religion is Jehovah’s witness. He has been suffered from the left painful thighs for several days and also experienced left-sided weakness. On arrival at the ER, he developed a generalized seizure. Consecutively, cardiac arrest occurred during preparation for brain imaging. Fortunately, the cardiac rhythm was recovered 3 minutes after cardiopulmonary resuscitation. However, hypoxia was not improved despite mechanical ventilation with high oxygen flow while his vital signs looked stable without any inotropic support. The level of hemoglobin (Hb) and hematocrit (Hct) was 15.8 g/dL and 46.8% respectively. There is mild cardiomegaly on chest X-ray. A chest computed tomography (CT) scan to evaluate the cause of sudden collapse revealed large sized filling defects in both main pulmonary arteries suggesting massive pulmonary thromboembolism (PTE) (Figure 1-A and B). We decided to apply PCPS using ECMO (Capiox EBS®, Terumo, Tokyo, Japan) for respiratory support and explained the need of ECMO to the patient’s guardian. They agreed with the ECMO procedure but strongly refused any type of blood transfusion according to his religious belief. Emergency veno-venous (VV) type ECMO was applied (Figure 2-A to C). There was no evidence of cardiovascular collapse at that time. Simultaneously performed coronary angiography showed chronic total occlusion at proximal to middle part of the left anterior descending artery with very good collateral flow from the distal part of the right coronary artery (Rentrop grading of II-III/III) (Figure 3-A and B). Immediately after ECMO procedure, the level of Hb and Hct decreased down to 11.7 g/dL and 36.5 % respectively. At the 3 days of hospitalization, his vital sign was very stable and global oxygenation was improved with minimal support by ECMO. However, the level of Hb and Hct was gradually decreased to 7.7g/dL and 23.6%. We inevitably decided the ECMO removal. Blood loss minimizing technique during decannulation should be attempted.
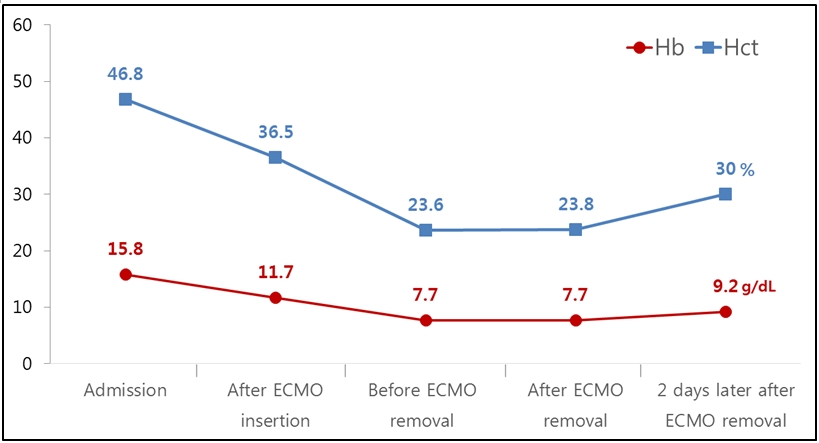
Therefore, we tried blood loss minimizing technique during decannulation as follows; after first venous access cannula was clamped, total 600cc of saline was continuously and slowly infused to the EMCO circuit including both venous cannulae, pump, and membrane chamber till the whole ECMO circuit was fulfilled with saline. Then, venous return cannula was clamped and both cannulae were carefully removed at 0 rpm of motor speed (Figure 4-A to F). The level of Hb and Hct was 7.7 g/dL and 23.8 % on ECMO removal day. Contrary to our expectation, it was increased up to 9.2 g/dL and 30.0 % respectively 2 days later after the circuit removal (Figure 5). There was no significant hemodynamic change including vital sign between before and after ECMO removal.
Conclusion
Although the additional risk of air and thrombus entering into the patient’s circulation and volume overload is still concerned, our blood loss minimizing technique with careful and continuous saline infusion into circuit during ECMO removal would be greatly helpful for critical patients who cannot accept blood transfusion due to any cause.
References
1. Russo MJ, Merlo A, Eton D, Patel PJ, Fedson S, Anderson A, Shah A, Jeevanandam V. Successful use of ECMO in a Jehovah's Witness after complicated re-heart transplant. ASAIO J. 2013;59(5):528-9.
2. Preston TJ, Olshove VF Jr, Chase M. Bloodless extracorporeal membrane oxygenation in the Jehovah's Witness patient. J Extra Corpor Technol. 2012;44(1):39-42.
3. Lindholm J, Palmér K, Frenckner B. Long-term ECMO treatment in Jehovah's Witness patient without transfusions. Perfusion. 2012;27(4):332-4.
4. Kleinman I. The right to refuse treatment: ethical considerations for the competent patient. CMAJ. 1991;144(10):1219-22.
5. Makdisi G, Wang IW. Extra Corporeal Membrane Oxygenation (ECMO) review of a lifesaving technology. J Thorac Dis. 2015;7(7):E166-76.
6. Kjærgaard B, Rasmussen BS, de Neergaard S, Rasmussen LH, Kristensen SR. Extracorporeal cardiopulmonary support may be an efficient rescue of patients after massive pulmonary embolism. An experimental porcine study. Thromb Res. 2012;129(4):e147-51.
7. Leick J, Liebetrau C, Szardien S, Willmer M, Rixe J, Nef H, Rolf A, Hamm C, Möllmann H. Percutaneous circulatory support in a patient with cardiac arrest due to acute pulmonary embolism. Clin Res Cardiol. 2012;101(12):1017-20.
 |
|
Figure 1-A and B, Chest computed tomography (CT) scan. Chest CT showed large sized filling defects in both main and lower lobe pulmonary arteries suggesting massive pulmonary thromboembolism (PTE).
|
 |
|
Figure 2-A, B, and C, Veno-venous extracorporeal membrane oxygenation (VV ECMO). The access cannula in the left femoral vein drained blood from the venous system into the ECMO circuit. The return cannula in the right femoral vein delivered blood back to the patient from the ECMO circuit.
|
 |
|
Figure 3-A and B, Coronary angiography. The left anterior descending artery with very good collateral flow from the distal part of the right coronary artery (Rentrop grading of II-III/III).
|
 |
|
Figure 4-A to F, Minimizing blood loss ECMO removal technique: The access cannula was clamped (yellow circle) after saline was connected at the access cannula line (white rectangles) (A and B). Total 600cc of saline was continuously and slowly infused to the EMCO circuit including both cannulae, pump, and membrane chamber till the whole ECMO circuit was fulfilled with saline (D). The return cannula was clamped and both cannulae were carefully removed at 0 rpm of motor speed. There was quite different color change in the circuit before (C and E) and after (D and F) the ECMO removal.
|
 |
|
Figure 5. Serial change of the level of hemoglobin (Hb) and hematocrit (Hct). No decreased of the level of Hb and Hct was noted at least.
|














